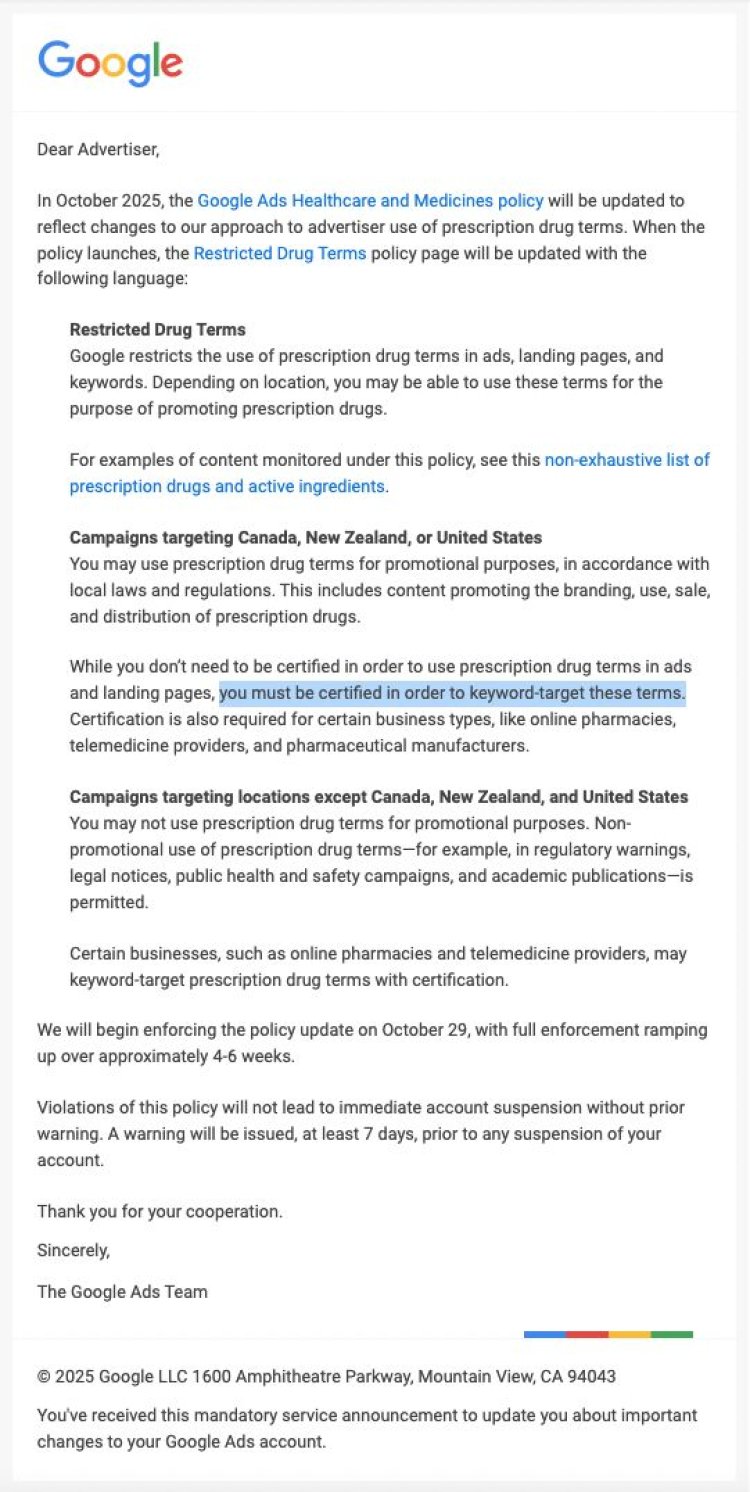
Google’s Bold Move: Certification Now Mandatory for Prescription Drug Advertisers
Google Ads introduces a sweeping change- effective October 29, advertisers must obtain certification to target prescription drug keywords, marking a major regulatory shift for healthcare, pharma, and legal marketing industries.

In a move sending shockwaves through the digital advertising world, Google Ads is tightening its policies on medical marketing. Starting October 29, advertisers bidding on prescription drug-related keywords will need to secure certification to continue running campaigns- marking one of the most significant overhauls in healthcare ad compliance in years.
This change directly affects a broad spectrum of advertisers, from pharmaceutical manufacturers and online pharmacies to telemedicine startups and legal firms running campaigns tied to pharmaceutical litigation (known as mass tort advertising).
A Global Policy Redefining Ad Boundaries
Under the new policy, Google will restrict the use of prescription drug terms in ads, keywords, and landing pages, with strict regional variations:
United States, Canada, and New Zealand:
Promotion of prescription drugs will remain permitted only for certified advertisers and businesses compliant with applicable local laws. Online pharmacies, telehealth providers, and pharma companies must obtain certification to bid on such keywords.
Everywhere else:
Using or promoting prescription drug-related terms is completely prohibited except in non-promotional contexts such as academic research, educational campaigns, public health communications, or legitimate government announcements.
The enforcement rollout begins October 29 and will be ramped up gradually over four to six weeks. Google has confirmed that advertisers will receive a seven-day warning before any account suspension is triggered.
Voices from the Ad Industry: Confusion and Concern
The update quickly ignited conversations across the marketing landscape. The reactions range from cautious to outright frustrated.
Anthony Higman, founder and CEO of AdSquire, was among the first to share Google’s notification publicly on LinkedIn, calling it a “pretty wild Google Ads update.” He said,
“You now must be certified to keyword-target prescription drug terms. Keywords used to be fair game- this is a big shift, especially for mass tort advertisers.”
Jyll Saskin Gales, a well-known Google Ads Coach, echoed industry confusion about how this change will affect keyword behavior, adding,
“Even if you avoid drug names directly, broad match or AI-driven Performance Max variations can still trigger those terms. How can advertisers control that?”
Meanwhile, Abby Nelson, a SEM strategist, called the move “ironic,” pointing to years of advertiser frustration:
“We used to need LegitScript certification just to mention prescription drugs on landing pages. Now broad match pull in drug terms like Botox- and ads are still getting approved.”
Compliance Becomes the Centerpiece of Healthcare Advertising
The overarching goal, Google says, is compliance and consumer protection. The certification requirement aims to reduce misuse of prescription-related keywords, fake pharmacy sites, and unverified telemedicine operations- issues that have plagued digital ad spaces for over a decade.
However, the update also means higher compliance costs, operational friction, and slower campaign approvals. Healthcare marketers, legal advertisers, and telehealth platforms will now need to integrate certification audits into their ad optimization workflows before running or updating campaigns.
This tightening of control signals that even keyword targeting- a core Google Ads function- is no longer exempt from regulatory scrutiny.
Why It Matters: A New Era for Healthcare Marketing
For healthcare marketers and agencies managing pharmaceutical or wellness clients, this is more than a routine update- it’s a reset of compliance expectations.
Legal advertisers running mass tort campaigns around drug side effects or recalls (e.g., Zantac or Ozempic) will likely see disruptions. Additionally, telemedicine startups like Cerebral, PlushCare, and Hims that rely heavily on Google Ads for visibility face new hurdles in keyword compliance.
The policy also reflects Google’s ongoing prioritization of user safety and crackdowns on questionable medical claims that could mislead consumers. According to Search Engine Land (owned by Semrush), this marks a move toward a “compliance-first” ecosystem where even search intent must meet certification guidelines.
The Bigger Picture: Google’s Expanding Compliance Web
This isn’t Google’s first attempt to regulate sensitive industries. It follows a history of verification systems- LegitScript certifications for online pharmacies, Google Ads Health Professional verifications, and even identity verification for political advertisers.
But what’s different now is scope. The new rule covers every point of campaign setup from keyword strategy to landing page compliance, redefining how advertisers structure their accounts.
As Google continues leveraging AI-driven ad matching and automation, marketers fear the unpredictability of triggers that could unintentionally violate these new rules. This will likely lead to increased reliance on certified third-party agencies or oversight software to maintain compliance.
The Bottom Line
Google’s new certification rule is more than a policy update- it’s a pivotal shift in digital healthcare advertising. By mandating certification to target drug-related keywords, the tech giant is signaling a new era of accountability, one that blurs the lines between regulation, automation, and brand responsibility.
For advertisers, it’s both a warning and an opportunity: evolve toward ethical, compliant, and transparent marketing- or risk getting caught in the tightening web of Google’s healthcare ad governance.












